
Clinical Rationale & Role
Clinical Rationale for Supplemental Imaging
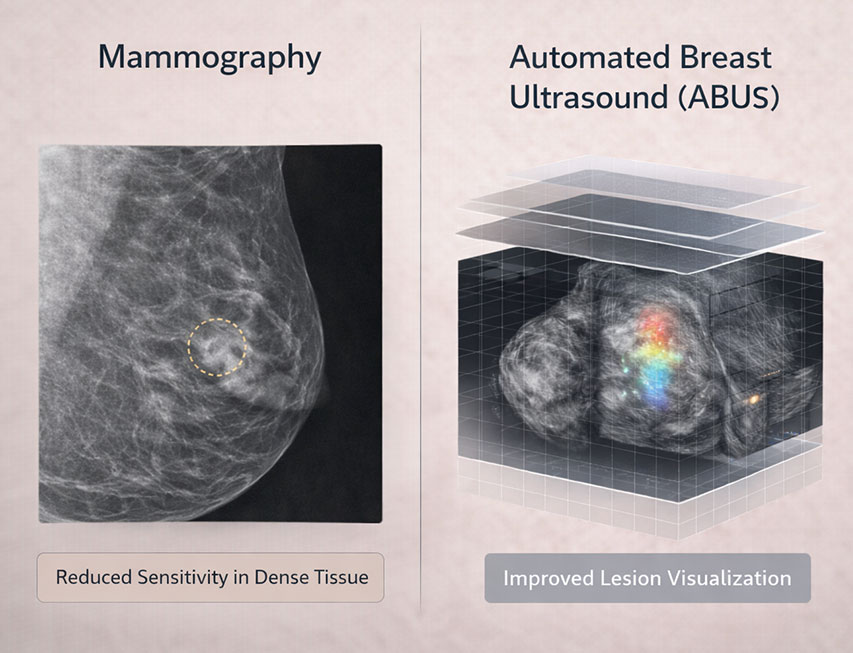
Mammography remains the gold standard for breast cancer screening and has well-established efficacy in reducing breast cancer mortality. However, its sensitivity is reduced in women with dense breast tissue, where dense fibroglandular tissue and malignancies can both appear radiopaque, potentially obscuring lesions on 2D mammograms.
Automated Breast Ultrasound (ABUS) is an FDA-cleared supplemental imaging technique specifically designed to address this limitation by providing standardized, reproducible, three-dimensional ultrasound imaging of the entire breast. It is intended to be used in conjunction with, not as a replacement for, mammography in asymptomatic women with dense breasts (ACR BI-RADS categories C or D).
Automated Breast Ultrasound (ABUS) is an FDA-cleared supplemental imaging technique specifically designed to address this limitation by providing standardized, reproducible, three-dimensional ultrasound imaging of the entire breast. It is intended to be used in conjunction with, not as a replacement for, mammography in asymptomatic women with dense breasts (ACR BI-RADS categories C or D).
How ABUS Works
Imaging Mechanism & Workflow
ABUS employs an automated wide-field transducer to acquire volume based, high-resolution 3D ultrasound data without operator-dependent variability inherent in handheld ultrasound (HHUS). The standardized acquisition provides axial, coronal, and sagittal reformats to support systematic review and interpretation.
This automation improves consistency and reproducibility relative to operator-dependent modalities and can be incorporated into radiology workflows with minimal additional personnel time.
Evidence Summary
Clinical Evidence & Diagnostic Performance
- Multiple studies demonstrate that adding ABUS to screening mammography in women with dense breasts increases cancer detection rates compared with mammography alone. For example, large cohort and meta-analyses have shown increases in detection of invasive cancers that might otherwise be occult on mammography.
- ABUS improves sensitivity in dense breasts, detecting additional cancers and small lesions that may not be visible on standard mammography.
- The SomoInsight study and related analyses support its role in supplemental screening to enhance lesion detection, particularly for invasive carcinoma.
- Our clinic uses advanced ultrasound and mammography technology to provide clear and accurate results.
Appropriate Use & Patient Selection
You may be a great fit for Genesis if:
- Asymptomatic women with dense breasts (BI-RADS C/D) following normal or benign mammographic screening.
- Women whose breast cancer risk profile includes dense tissue recognized as a factor lowering mammographic sensitivity.
- Patients for whom enhanced visualization is desired due to breast density or clinical risk factors, including family history when combined with standard screening regimes.
Note: ABUS is intended to supplement mammography when indicated and is not recommended as a stand-alone screening tool in routine practice absent specific clinical justification.
Integration into Clinical Workflow
Referral & Interpretation Considerations

- Referrals for ABUS should include documentation of breast density (ACR BIRADS) and relevant clinical risk factors.
- ABUS exams typically take 15–25 minutes and produce standardized 3D datasets that can be reviewed efficiently by trained breast imagers
- Comparing current and prior imaging enhances interpretation accuracy; radiologists should be provided with all available mammography and supplemental imaging data.
Safety Profile
Safety & Patient Tolerance
ABUS uses non-ionizing sound waves and does not expose patients to radiation. The absence of ionizing radiation makes it a low-risk supplemental modality appropriate for repeated use when indicated. The exam procedure is generally well tolerated, with patients reporting minimal discomfort and no need for breast compression.
Genesis Screening Model
How Genesis Applies ABUS Clinically
At Genesis Breast Screening, we employ ABUS in symptomatic women with dense breast tissue as part of a supplemental screening strategy designed to enhance detection sensitivity and povide referring clinicians with comprehensive imaging. Our protocols incorporate consistent acquisition, standardized interpretation, and integration with mammographic findings to support clinical decision-making.
Summary Points for Referring Providers
Key Takeaways

ABUS is the only FDA-approved supplemental ultrasound screening technology for women with dense breasts.

ABUS adds diagnostic sensitivity but may increase recalls and biopsies; clinical judgment is essential.

Evidence supports enhanced cancer detection when used in conjunction with mammography in appropriate populations.

Integration into practice requires breast density reporting and appropriate case selection.
References & Further Reading
Links / Citations for Providers
- Dense breast imaging background and supplemental strategies. National Library of Medicine
- Invenia ABUS technology overview and FDA clearance context. GE HealthCare Invenia ABUS Premium
- Comparative performance studies. PubMed
- Extended clinical evidence and diagnostic performance. PubMed

